Search
News & Events
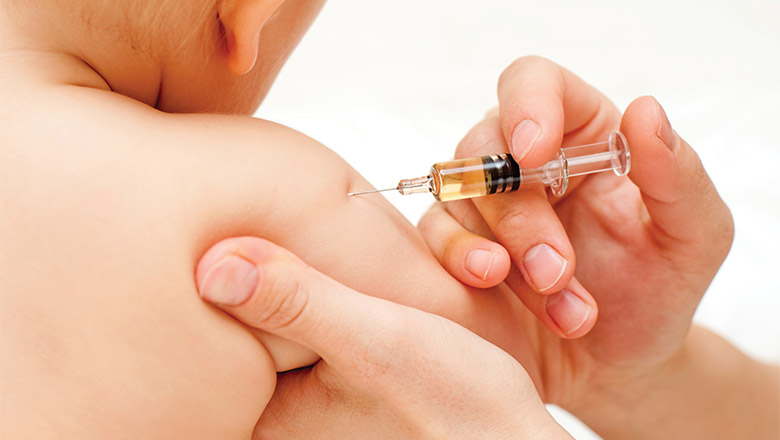
Study to protect babies from flu as flu season strikes earlyParents of babies in Perth and Adelaide are being urged to take part in a landmark study to examine the best ways to keep their child safe from influenza this winter, amid a surge in serious infections.

News & Events
Childhood influenza vaccination rates improves with better accessMore children across Australia are being vaccinated against the flu since funding was expanded and access widened under the National Immunisation Program

Wrap up of the people, projects and updates from 2017 in the Vaccine Trials Group research area, and Dr Peter Richmond.
Contact us If you'd like to get in touch, please contact us by phone or email. Phone: 0400 450 240 Email: vtg@thekids.org.au Respiratory Syncytial

Meningococcal disease is caused by the bacteria Neisseria meningitidis, or 'meningococcus'. It's an uncommon but very serious disease that can result in death.
The Wesfarmers Centre is pleased to announce the successful recipients for the 2021 Round 2 Seed Funding Grants. Julie Hibbert | Validating a

News & Events
Latest research identifies true danger of antimicrobial resistance in Australian kidsOne out of every 10 children with a bloodstream infection are infected with a multi-drug resistant organism in the nation’s first-ever surveillance study investigating the prevalence of paediatric antimicrobial resistance (AMR).

News & Events
Latest infectious disease guidelines aim to keep childcare ‘bug-free’The National Health and Medical Research Council (NHMRC) has launched their sixth edition of Staying healthy: Preventing infectious diseases in early childhood education and care services in a bid to tackle the transmission of germs amongst young kids.

News & Events
Major funding aims to improve skin cancer outcomes for young Aboriginal peopleVital research promoting sun smart choices and skin cancer prevention for young Aboriginal people is now underway at The Kids Research Institute Australia thanks to a $100,000 Perpetual 2024 IMPACT Philanthropy grant.

News & Events
The Kids researchers granted $5 million to prevent RHD across PacificA team led by Dr Joseph Kado from the Wesfarmers Centre of Vaccines and Infectious Diseases, based at The Kids Research Institute Australia, and The University of Western Australia (UWA) has been awarded $5 million by the Federal Government in a major push to prevent rheumatic heart disease across the Pacific.
